
For decades, immune-based cancer therapies have offered hope but also risk, with powerful tumor-fighting effects often shadowed by dangerous side effects. Now, a new approach is emerging that could change the equation: natural killer (NK) cell therapies are showing early clinical results that combine potent cancer targeting with an unprecedented safety profile.
The Body’s Innate Defenders
NK cells are the immune system’s rapid responders, capable of attacking cancer cells without prior training or genetic matching. Unlike T cells, which require customization for each patient, NK cells can be used “off the shelf,” making them easier to produce and distribute. This simplicity has fueled a surge in research, with the number of clinical trials involving NK cell therapies rising from just 19 in 2020 to nearly 90 by late 2024. Scientists are now exploring how to harness these innate defenders to deliver precise, scalable cancer treatments.
A Safer Alternative to CAR-T
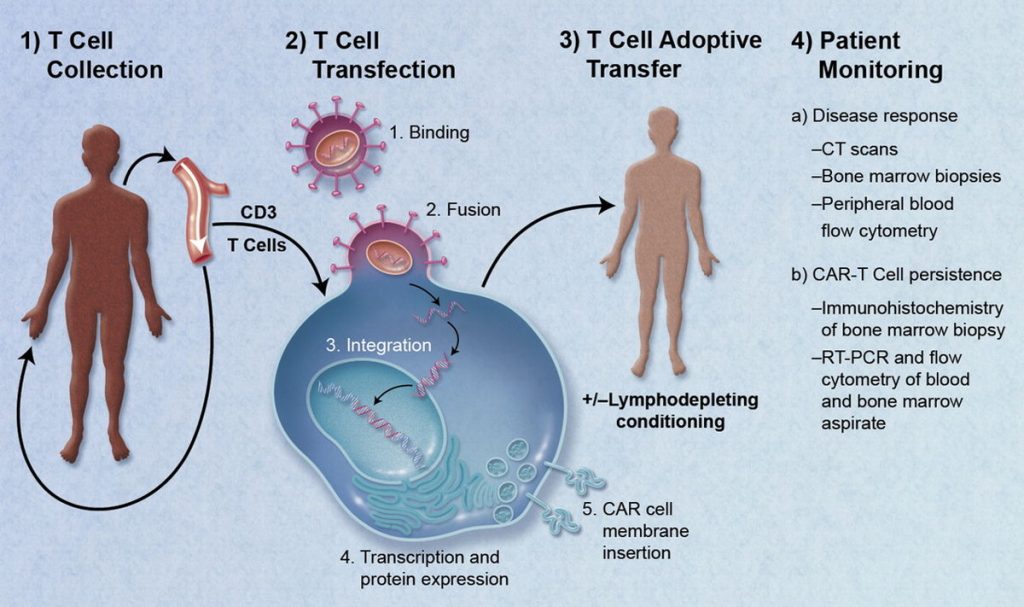
Chimeric antigen receptor T-cell (CAR-T) therapy revolutionized cancer care, especially for blood cancers, but its benefits have come with significant risks. Patients often face severe immune reactions, including cytokine storms and neurological complications, requiring intensive hospital monitoring. In contrast, NK cell therapies have demonstrated a remarkable safety record. A review of 31 clinical trials found only mild side effects, such as headaches or fatigue, and no cases of life-threatening immune reactions. In one leukemia study, only a single patient experienced a mild fever, easily managed with standard care. For oncologists accustomed to the risks of CAR-T, these results mark a significant shift.
Breakthroughs Across Cancer Types

Early clinical trials are delivering promising results across a range of cancers. In a study of patients with Waldenstrom macroglobulinemia, a rare and treatment-resistant blood cancer, two individuals achieved complete remission after NK cell therapy—one with NK cells alone, the other with a combination of NK cells and rituximab. Both were treated as outpatients and experienced no severe side effects.
Acute myeloid leukemia patients have also seen encouraging outcomes. In a phase 1 trial, six out of ten participants achieved complete remission within four weeks, with some remaining cancer-free for over eight months. Minimal side effects were reported, and several patients proceeded to potentially curative bone marrow transplants.
The SENTI-202 CAR-NK trial, presented at the 2025 AACR conference, highlighted the potential of advanced NK cell engineering. Among nine patients with relapsed or refractory leukemia, nearly half achieved complete, ongoing remission, with no dose-limiting toxicities. The therapy’s “logic-gate” design enabled it to distinguish cancer cells from healthy ones, sparing normal tissue and reducing risk.
Expanding the Reach: Solid Tumors and Beyond
While blood cancers have been the primary focus, NK cell therapies are making inroads against solid tumors—a notoriously difficult target for immunotherapy. In one study, 12 lung cancer patients with EGFR mutations received NK cells combined with cetuximab. All participants experienced either tumor stability or reduction, with one in four seeing measurable shrinkage and no severe side effects.
Liver cancer trials have reported a 72% objective response rate, far surpassing existing immunotherapies. Combining NK cells with local treatments like ablation or surgery further improved outcomes without increasing risk. In ovarian cancer, researchers at Dana-Farber are testing “memory-like” NK cells, engineered to remember and attack tumors more effectively. Early results show encouraging safety and engagement, offering hope for patients with few other options.
Engineering for Endurance and Precision
Scientists are enhancing NK cell therapies by equipping them with cytokines like IL-15, which help the cells survive and remain active longer. Trials of IL-15–engineered CAR-NK cells have achieved 64% complete remission rates, with the cells persisting for up to a year—reducing the need for repeated treatments.
The FDA’s 2024 approval of ANKTIVA, an IL-15 receptor agonist, for bladder cancer patients unresponsive to standard therapy, marked a milestone for NK cell science. In the pivotal study, median complete responses lasted nearly four years, with side effects in less than 3% of patients.
Looking Ahead: Accessibility and the Future of Cancer Care
Beyond safety and efficacy, NK cell therapies offer the promise of affordability and accessibility. Unlike CAR-T, which can cost hundreds of thousands of dollars and take weeks to manufacture, NK therapies can be mass-produced in days and do not require genetic matching. This could make advanced cancer immunotherapy available to a broader population, including patients in countries with limited healthcare resources.
With nearly 90 clinical trials underway and expanding applications across cancer types, NK cell therapies are poised to reshape the landscape of cancer treatment. Their ability to deliver powerful, targeted responses without the severe side effects of earlier immunotherapies signals a new era—one where survival does not come at the cost of safety or dignity. As research continues, the hope is that NK cells will not only complement but, in some cases, replace existing therapies, bringing effective and accessible cancer care to more patients worldwide.